REPORTS
Biologics and Psoriasis: the Beat Goes On
Presented by: Mark Lebwohl, MD, FAADWaldman Professor and Chairman, Kimberly and Eric J. Waldman Department of Dermatology, Icahn School of Medicine at Mount Sinai, New York, NY, USA
While there are a number of biologics available for psoriasis, each have different characteristics that make one more useful than another in a specific patient population and/or rationale.
Speed
In terms of speed, defined as time to achieve 50% improvement in baseline PASI scores, brodalumab and ixekizumab showed response in as early as 2 weeks post dose. High-dose secukinumab, adalimumab, and infliximab show a response within 3 to 4 weeks, while ustekinumab after 4 weeks. Instead, etanercept present a response after 6 and 11 weeks with high-dose and low-dose, respectively.1
Durability
- Secukinumab delivers high and long-lasting skin improvement, which has been demonstrated over a 5-year period.1 There was an approximately 90% PASI 75 responder rate observed by year 1 that was maintained for a 5-year period.
- Ixekizumab has also demonstrated PASI 50 in more than 60% of patients by week 12.2 By the end of a 60-week study, 53% of patients maintained a PASI 50 while 38% kept a PASI 75.
- Brodalumab has shown high percentages of PASI 75 and PASI 90 over a 5-year period with a PASI 75 of approximately 90% and PASI 90 of about 70%.3
- The VOYAGE 1 study demonstrated a PASI 90 of 80% to 82% over a 100-week period with the use of guselkumab and a PASI 100 of 49% to 55%.4
- Lastly, risankizumab also demonstrated 66% of patients who maintained a PASI 100 for 41 to 66 weeks.5
Safety
Etanercept and adalimumab have both demonstrated long-term safety. In detail, etanercept demonstrated safety of up to 10 years while adalimumab demonstrated long-term safety in 23,458 patients with rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, psoriatic arthritis, psoriasis, and Crohn’s disease.6 In addition, TNF blocker usage is associated with a lower incidence of cardiovascular events in patients with rheumatoid arthritis and psoriasis.7-10
Pregnancy
Certolizumab is the only biologic safe for use in pregnancy as it lacks the Fc portion and thus cannot cross the placenta. In a study focusing on the concentration of certolizumab in the blood of mothers and cord blood of infants (n=14 mother/infant pairs), only one infant had detectable levels of 0.040 µg/mL in the umbilical cord.11
Use in Obese Patients
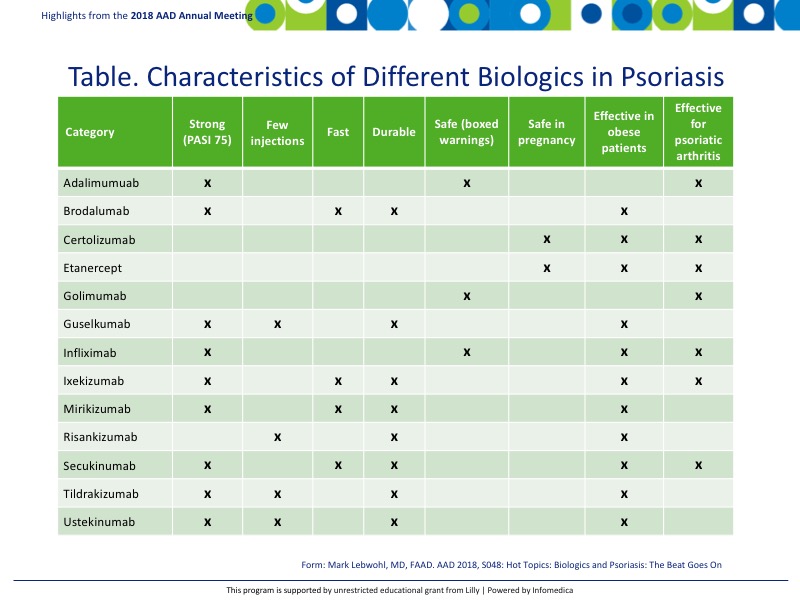
Many of the current biologics are effective in patients who are obese. Some may require dose adjustments for weight while others do not (Table).
Use in Psoriatic Arthritis
There are a few main biologics that have shown effectiveness in psoriatic arthritis including:
- Etanercept
- Adalimumab
- Infliximab
- Certolizumab
- Golimumab
- Secukinumab
- Ixekizumab
- Ustekinumab (not as effective as the previous biologics).
See Table.
Key Messages
- There are many different characteristics of biologics that should be considered based on the needs of the patient and the remediation for their disease.
- Having a better sense of these biologics will allow for better options and improved outcomes.
REFERENCES
Present disclosure: The presenter has reported that he receives research funds from Abbvie, Amgen, Boehringer Ingelheim, Celgene, Eli Lilly, Janssen/Johnson & Johnson, Kadmon, Medimmune/Astra Zeneca, Novartis, Pfizer. Henis also a consultant for Allergan, Leopharma, and Promius.
Written by: Debbie Anderson, PhD
Reviewed by: Victor Desmond Mandel, MD
